ABOUT US
TELEFONO E FAX
CONTATTI
INDIRIZZO
Zeta Research S.r.l.
P.I. 01028800322
N° REA TS - 118955
Via A. Caccia, 8
Trieste
34129
Italia
Zeta Research S.r.l. è conforme ai requisiti UNI EN ISO 9001:2015 - N. di certificazione: 50 100 10601 - Rev. 007
Clicca qui per visualizzare il certificato
Clicca qui per visualizzare l'informativa privacy
Zeta Research Srl © 2024
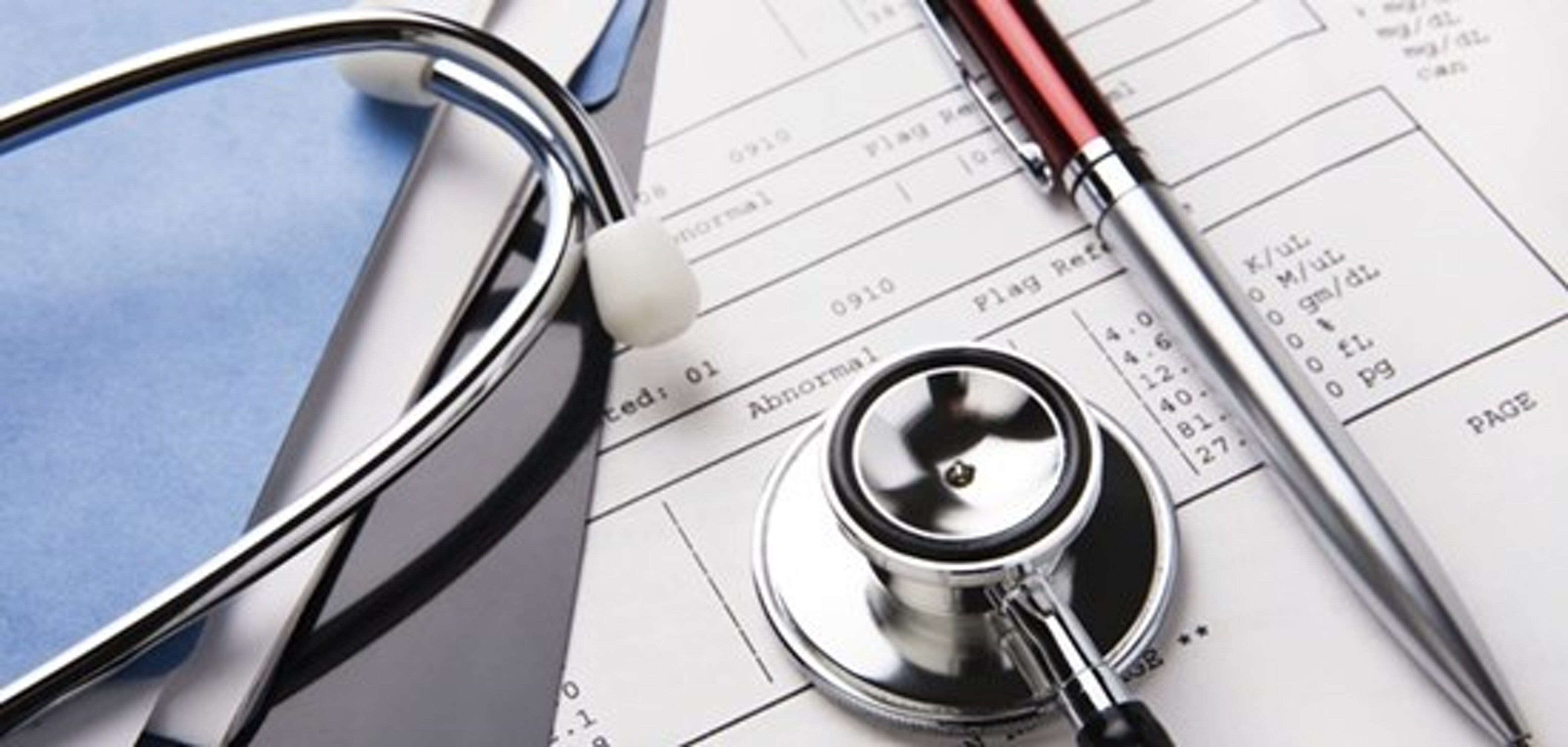
CLINICAL TRIALS AND EPIDEMIOLOGICAL STUDIES
Zeta Research offers a complete service that follows clinical and pharmaceutical research in the following sectors: medicine, biomedicine, nutrition, biotechnology, and medical devices.
Clinical planning and management
- Administrative authorizations;
- Cost-efficiency and risk-benefit analysis;
- Documentation for Ethics Committees (EC);
- Methodological and normative adequacy;
- Notification to the Competent Authorities;
- Process control and follow-up;
- Protocol drafting;
- Research planning;
- Site recuitment;
- Start-up clinical experimentations;
- Submission to the Ethics Committees;
- Treatment allocation.
Data Collection & Data Management
- Breaking the blind;
- Data Collection;
- Data Entry;
- Design and realization of Case Rerport Form;
- ITC (Information and Communication Technologies) solution for randomizing;
- Management randomization;
- Medical coding;
- Transfer and data encryption;
- Validation and verification of data.
Statistical and Metodological Consulting
- Endpoints identification and quantification;
- Sample design and size;
- Statistical analysis;
- Statistical Analysis Plan;
- Statistical Metodology design;
- Statistics sections for the clinical study protocol;
- Study design choice.
Medical writing and reporting
- Development Safety Update Report (DSUR) drafting;
- Scientific papers;
- Scientific posters;
- Study reporting;
- Technical document writing and management, both for Medical Devices EC marking or medicines registration (Clinical Evaluation Report, Expertise, Position Paper and Risk Analysis) and for clinical studies and post-marketing evaluation of effectiveness and security (product commonly used in the clinical practice).
ABOUT US
PHONE AND FAX
CONTACTS
ADDRESS
Zeta Research S.r.l.
P.I. 01028800322
N°REA TS - 118955
Via A. Caccia, 8
Trieste
34129
Italia
Zeta Research Srl has been found to comply with the requirements of UNI EN ISO 9001:2015 - Certification No: 50 100 10601 - Rev. 007 .
Click here to view the certificate
Click here to view the Privacy Information
Zeta Research Srl © 2024
